藥品短缺可能會影響病人照護的品質及用藥選擇,即使有替代品也可能會衍生新的問題,如治療效果與副作用的改變、醫療人員對新品項的不熟悉造成用藥疏失等,進而增加醫療成本,故對公眾健康福祉有潛在重大的影響。
為降低藥品供應短缺可能的衝擊,衛生福利部食品藥物管理署自100年起委託社團法人臺灣臨床藥學會執行藥品供應短缺處理連續型計畫,建置「主管機關、各級醫療機構、製藥業及相關公協學會」等單位共同處理藥品短缺的問題之模式及合作平台,具體方法則是透過建置藥品供應資訊平台、建構藥品短缺處理機制、訂定臺灣必要藥品清單、盤點緊急連絡網、舉辦工作小組暨專家會議及藥師共識會議、舉辦藥師教育宣導課程及參與跨部門溝通小組等七大面向提升我國處理相關事件之效能,以減緩藥品短缺造成的衝擊,並保障民眾用藥權益。
計畫執行團隊成員來自臺灣北、中、南、東四區,包括北榮、臺大、新光、林口長庚、高雄長庚、中醫大、花蓮慈濟等七家醫學中心之藥事部門主管、專科臨床藥師與藥品供應專責藥師等約60名專業藥師,並設有專人監控我國藥品供應資訊平台及先進國家藥品短缺網站上之相關訊息。平台接收到藥品短缺之通報訊息後,會由各醫學中心輪值之專業臨床藥師針對通報藥品進行評估,評估重點包括該藥品供應實況、短缺時程、短缺原因,並根據藥品成份、劑量、劑型與臨床使用適應症尋找替代藥品,主管機關也會衡量藥品臨床必需性及緊急度裁決是否需啟動「公開招商、庫存調查、專案進口或專案製造」等機制,必要時更會展開跨司署的合作,協請健保署及醫事司共同處理藥品短缺問題。
當藥品供應產生問題時,可能需要使用與原先不同的藥品,不論替代品是原廠藥或學名藥,國內主管機關皆會嚴密把關監測品質,以確保其療效及安全性。專業醫療人員面對藥品供應問題時,應秉持正向態度教育民眾協助建立正確用藥觀念,並提醒民眾切勿擅自停藥,以降低藥品變化對治療成效的影響。
Drug shortages may affect the quality of patient care and treatment options. Even alternative products are available, they may cause other problems, such as different efficacy and side effects or the potential for medical errors due to unfamiliar with alternative products. They also increase costs to hospitals and health care systems, and pose a great influence to the public’s health.
The Taiwan Food and Drug Administration (TFDA) has authorised the Taiwan Society of Health-System Pharmacists to conduct the relevant projects since 2011. The purpose of this project is to establish a multidisciplinary management flowchart to aid government agencies, health care institutions, pharmaceutical manufacturers, and pharmacist and pharmaceutical societies and associations in handling drug shortages. This project comprises seven phases: establishment of an online Taiwan drug supply database platform, stipulation of strategic plans for drug shortages, proposal for a Taiwan Essential Medication List, creation of an emergency inventory liaison network, committee and pharmacist consensus meetings to ensure internal consistency during evaluation, educational forums for pharmacists, and establishment of a cross-department communication team for special cases. Through these measures, impact from drug shortages can be relieved and patient safety in Taiwan can be improved greatly.
The task force covers the northern, central, eastern, and southern regions of Taiwan and contains approximately 60 members, including pharmacy directors, clinical pharmacists, and specialised drug supply pharmacists from seven medical centers, namely Taipei Veterans General Hospital, National Taiwan University Hospital, Shin Kong Wu Ho-Su Memorial Hospital, Chang Gung Memorial Hospital (Linkou and Kaohsiung Branches), China Medical University Hospital, and Hualien Tzu Chi Hospital. Project assistants monitor Taiwan drug supply database platform and global drug shortage information. When the drug supply database platform receives the drug shortage report, the review and verification process is initiated. Pharmacists at seven medical centers are assigned to initially review the report, confirm drug supplies in the real world, and further evaluate possible drug substitutes according to generic names, dosage form, strength, and indication. Other information, including urgency levels and clinical suggestions, is still required. If no substitute is available and the drug in short supply is required for clinical practice, the TFDA engage in drug shortage management to check the inventory and seek public suppliers or special import availability. If necessary, the TFDA discusses management strategies with relevant units, such as the National Health Insurance Administration (NHIA) or Department of medical affairs, Ministry of Health and Welfare (MOHW).
When drug shortages occur, we may need to use alternative products. No matter they are brand or generic drugs, the TFDA will monitor drug quality strictly. Healthcare professionals should remind patients to take medication regularly and keep a positive attitude when discussing with patients.
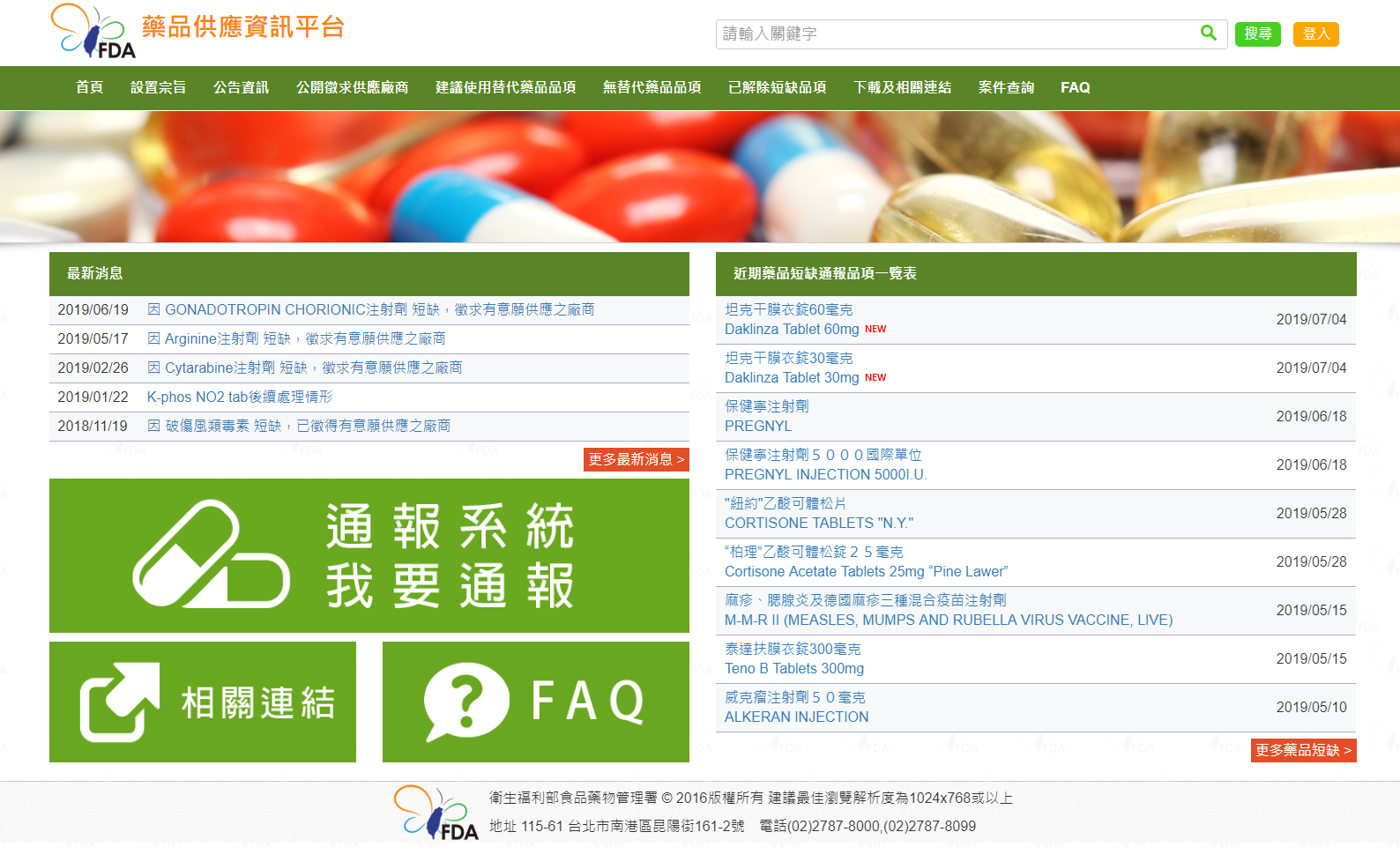
圖:我國藥品供應資訊平台
Figure. Taiwan drug supply database platform